MAGIS PHARMA
MAGIS PHARMA, THE MERGER OF FSA, FRAVER AND QUALENICA



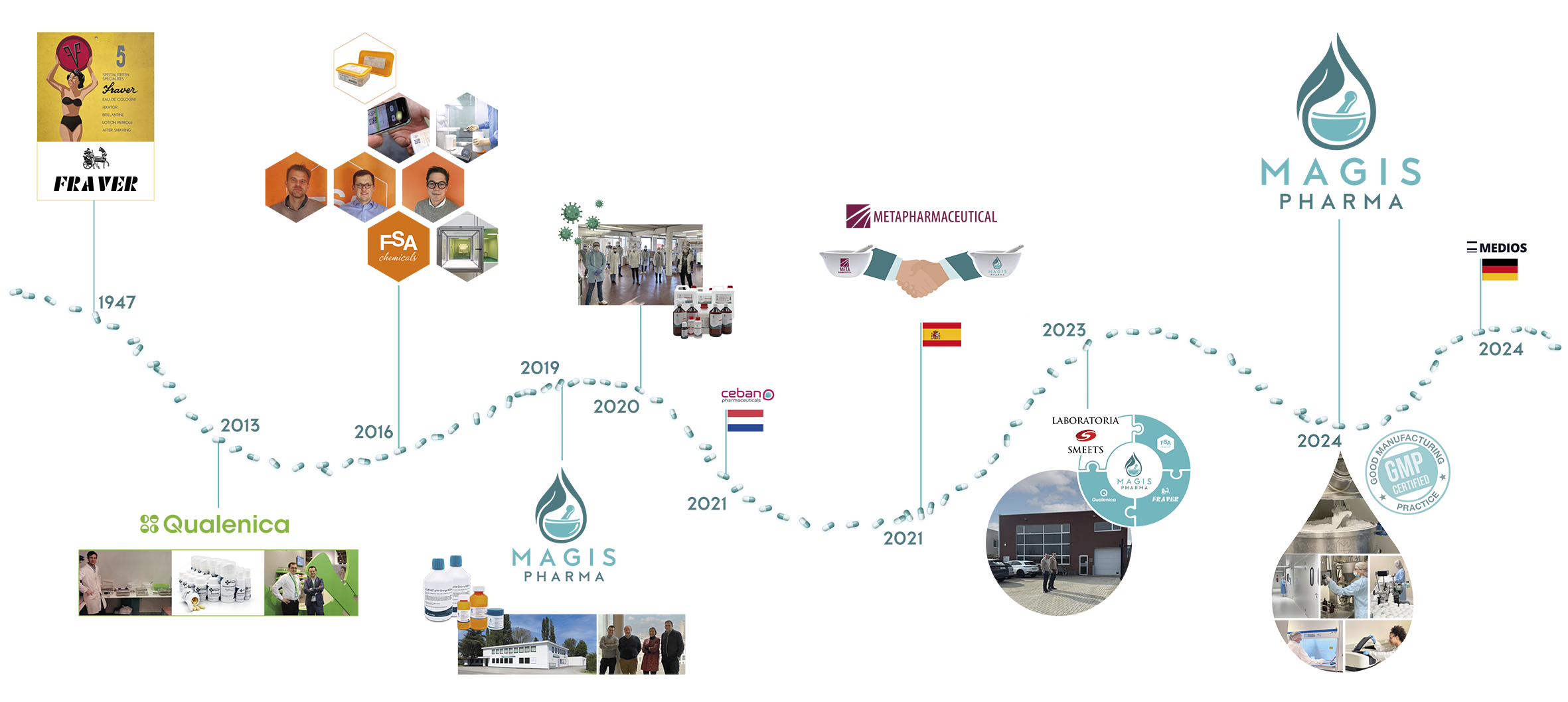
The Magis Pharma Group is the result of the merger between FSA‑Chemicals, Fraver and Qualenica. These three companies, which had already been collaborating for many years, saw the time as right to integrate so that they could work more efficiently and effectively. In 2023, Magis Pharma acquired Labo Smeets and moved all production to Wilrijk. In addition, Magis Pharma also distributes raw materials under the Pannoc brand. For pharmacies, the point of contact has now become a single organisation that can better respond to customer needs. Magis Pharma is part of a network of European companies specialised in pharmaceutical compounding, led by the Dutch compounding pharmacy CEBAN. Within this group, Magis Pharma focuses specifically on raw materials for compounding. Ceban Pharmaceuticals and Magis Pharma are part of Medios.
Why choose Magis Pharma?

Highly competitive prices
Because we guarantee the best price and apply a clear discount policy, you save both time and money, allowing you to work on your preparations with peace of mind.

Formulation support
The roles have now been reversed - you are the one asking questions to Professor Kligge! Vdk hosts the most visited formulation helpdesk in Belgium.

For and by compounders
Magis Pharma was founded by Belgium’s largest raw‑materials supplier, Qualenica. You benefit from our volume advantages. Our products are optimised for practical use.
About Magis Pharma
Our Production Sites in Compliance with GMP Standards
Within the group, we have two production sites that meet GMP requirements. At FASC, for example, we produce infusions, vials, ampoules (for injections and powders - Whitmount & exclusivity), while in Houten raw materials are processed into products and commercial packaging. What is it like to be part of the team?

Quality Control
A key aspect of our organisation is continuous quality control from the beginning to the end of every production cycle. To ensure the safe handling of a complete portfolio of pharmaceutical substances, our quality‑assurance team oversees the quality control of raw materials and reactions.

Professional and Flexible Logistics for Fast Delivery
To support the strong growth of the company, all finished products are picked externally. This takes place at several distribution platforms across Europe.

Discover our job opportunities
